Program Highlights:
-
AC302 was a successful pivotal Phase 3 trial across all three co-primary endpoints
-
Consistent results across AC301 and AC302 for two of three co-primary endpoints
-
Positive B-SIMPLE4 results validated our technology and our effective trial execution
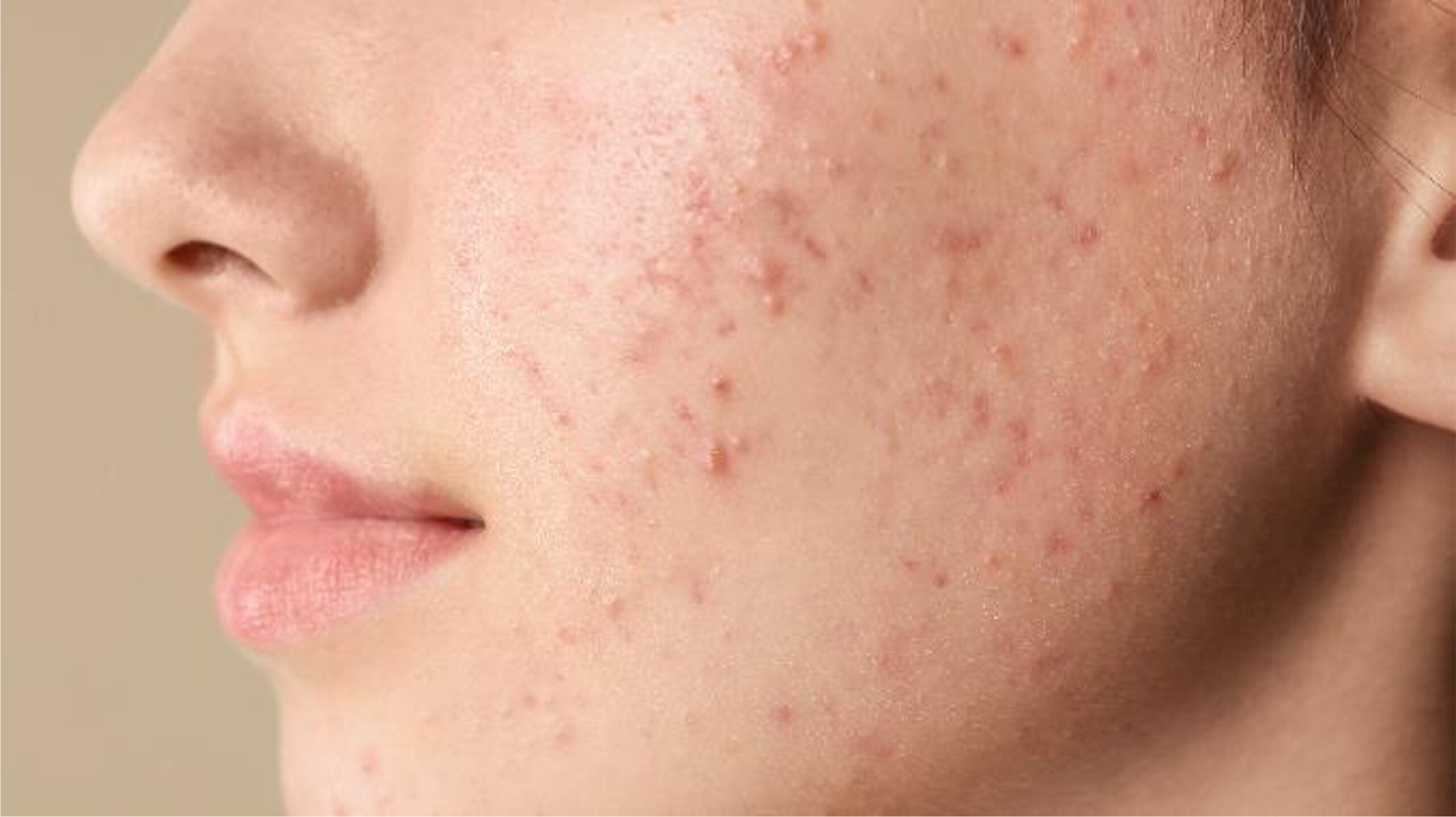
Acne Market
We believe among dermatology health-care providers there remains a significant unmet need for novel mechanisms of action with favorable tolerability profiles to improve patient outcomes in acne.
~50 million
People with
acne in the U.S.3
~$2.9 billion
Topical U.S. acne market (products
and combos)4
Acne has significant physical and psychological morbidity, such as permanent scarring, poor self-image, depression and anxiety
1. Vasam M, Korutla S, Bohara RA. Acne vulgaris: A review of the pathophysiology, treatment, and recent nanotechnology based advances. Biochem Biophys Rep. 2023;36:101578. Published 2023 Nov 23. doi:10.1016/j.bbrep.2023.101578 2. Baldwin et al Dermatol Ther. 2019;9(4):725–734 3. Bickers DR, Lim HW, Margolis D, Weinstock MA, Goodman C, Faulkner E et al. The burden of skin diseases: 2004 a joint project of the American Academy of Dermatology Association and the Society for Investigative Dermatology. Journal of the American Academy of Dermatology 2006;55:490-500. 4. IMS National Prescription Audit MAT Feb. 2016 – Acne Prescription Market TRx Dollars – MAT Feb 2016.